Seznamy Atom Vs Molecule Size Čerstvý
Seznamy Atom Vs Molecule Size Čerstvý. Every atom is made up of a nucleus and one or more electrons attached to it. Combination of two or more atoms. One or more protons and a number of neutrons make up the nucleus. The smallest particle with properties of an element. It is the smallest part an element can divide into.
Nejchladnější Molecular Size An Overview Sciencedirect Topics
It is the smallest part an element can divide into. Both are tiny units, but as molecules are made up of atoms, the size of an atom is much smaller than a molecule. Atoms are the tiny particles that make the whole world. The covalent radius of a chlorine atom, for example, is half the distance between the nuclei of the atoms in a cl 2 molecule.A molecule is made up of atoms that are bound by ionic, covalent or metallic bond.
The covalent radius of a chlorine atom, for example, is half the distance between the nuclei of the atoms in a cl 2 molecule. The covalent radii of the main group elements are given in the figure below. The size of an atom can be estimated by measuring the distance between adjacent atoms in a covalent compound. The smallest particle with properties of an element. It is the smallest part an element can divide into. Not just it, but like everything else too—planets, stars, universe in short. Moreover, to form ions, atoms gain or lose electrons, which is … Every atom is made up of a nucleus and one or more electrons attached to it.

Every atom is made up of a nucleus and one or more electrons attached to it. An atom may not always be stable … This article will study atoms, molecules and ions, the difference between atom and molecule and molecular elements in detail. The living portion of our world represent biology. Two or more atoms chemically bonded together. Not just it, but like everything else too—planets, stars, universe in short.. It is the smallest part an element can divide into.

The living portion of our world represent biology.. It is the smallest part an element can divide into. The covalent radii of the main group elements are given in the figure below. Most fundamental and smallest part that can exist of an element. One or more protons and a number of neutrons make up the nucleus. Combination of two or more atoms. A molecule is made up of atoms that are bound by ionic, covalent or metallic bond. It is the smallest part an element can divide into.

An atom may not always be stable … The covalent radii of the main group elements are given in the figure below. One or more protons and a number of neutrons make up the nucleus.. An atom may not always be stable …

Not just it, but like everything else too—planets, stars, universe in short. An atom may not always be stable … A molecule is made up of atoms that are bound by ionic, covalent or metallic bond. Not just it, but like everything else too—planets, stars, universe in short.

Atoms are the tiny particles that make the whole world. Atoms molecules and ions atoms and molecules definition. An atom may not always be stable … This article will study atoms, molecules and ions, the difference between atom and molecule and molecular elements in detail. The smallest particle with properties of an element... This article will study atoms, molecules and ions, the difference between atom and molecule and molecular elements in detail.

Two or more atoms chemically bonded together.. The covalent radii of the main group elements are given in the figure below. The size of an atom can be estimated by measuring the distance between adjacent atoms in a covalent compound. Not just it, but like everything else too—planets, stars, universe in short. A molecule is made up of atoms that are bound by ionic, covalent or metallic bond. Every atom is made up of a nucleus and one or more electrons attached to it. An atom may not always be stable … Atoms molecules and ions atoms and molecules definition... It is the smallest part an element can divide into.

Most fundamental and smallest part that can exist of an element. Moreover, to form ions, atoms gain or lose electrons, which is … The covalent radii of the main group elements are given in the figure below. The covalent radius of a chlorine atom, for example, is half the distance between the nuclei of the atoms in a cl 2 molecule. Every atom is made up of a nucleus and one or more electrons attached to it. The living portion of our world represent biology. It is the smallest part an element can divide into. Two or more atoms chemically bonded together.. One or more protons and a number of neutrons make up the nucleus.

It is the smallest part an element can divide into. . Combination of two or more atoms.

This article will study atoms, molecules and ions, the difference between atom and molecule and molecular elements in detail. . Every atom is made up of a nucleus and one or more electrons attached to it.

The covalent radius of a chlorine atom, for example, is half the distance between the nuclei of the atoms in a cl 2 molecule.. The size of an atom can be estimated by measuring the distance between adjacent atoms in a covalent compound. Both are tiny units, but as molecules are made up of atoms, the size of an atom is much smaller than a molecule. Atoms molecules and ions atoms and molecules definition. This article will study atoms, molecules and ions, the difference between atom and molecule and molecular elements in detail. Combination of two or more atoms.. The covalent radius of a chlorine atom, for example, is half the distance between the nuclei of the atoms in a cl 2 molecule.

The size of an atom can be estimated by measuring the distance between adjacent atoms in a covalent compound... Two or more atoms chemically bonded together. The smallest particle with properties of an element. Atoms molecules and ions atoms and molecules definition. Not just it, but like everything else too—planets, stars, universe in short.. The size of an atom can be estimated by measuring the distance between adjacent atoms in a covalent compound.

The covalent radii of the main group elements are given in the figure below.. A molecule is made up of atoms that are bound by ionic, covalent or metallic bond. The size of an atom can be estimated by measuring the distance between adjacent atoms in a covalent compound. Both are tiny units, but as molecules are made up of atoms, the size of an atom is much smaller than a molecule. Atoms are the tiny particles that make the whole world. Atoms molecules and ions atoms and molecules definition. This article will study atoms, molecules and ions, the difference between atom and molecule and molecular elements in detail. An atom may not always be stable … Most fundamental and smallest part that can exist of an element. It is the smallest part an element can divide into. Moreover, to form ions, atoms gain or lose electrons, which is … Both are tiny units, but as molecules are made up of atoms, the size of an atom is much smaller than a molecule.
Not just it, but like everything else too—planets, stars, universe in short. One or more protons and a number of neutrons make up the nucleus. Not just it, but like everything else too—planets, stars, universe in short. Moreover, to form ions, atoms gain or lose electrons, which is … The covalent radius of a chlorine atom, for example, is half the distance between the nuclei of the atoms in a cl 2 molecule. The living portion of our world represent biology... The smallest particle with properties of an element.

It is the smallest part an element can divide into... Not just it, but like everything else too—planets, stars, universe in short. Every atom is made up of a nucleus and one or more electrons attached to it.. The size of an atom can be estimated by measuring the distance between adjacent atoms in a covalent compound.

Atoms molecules and ions atoms and molecules definition. This article will study atoms, molecules and ions, the difference between atom and molecule and molecular elements in detail. The size of an atom can be estimated by measuring the distance between adjacent atoms in a covalent compound. Atoms are the tiny particles that make the whole world. Both are tiny units, but as molecules are made up of atoms, the size of an atom is much smaller than a molecule. One or more protons and a number of neutrons make up the nucleus. The living portion of our world represent biology. The covalent radii of the main group elements are given in the figure below. Combination of two or more atoms. Atoms are the tiny particles that make the whole world.
Every atom is made up of a nucleus and one or more electrons attached to it... One or more protons and a number of neutrons make up the nucleus. Moreover, to form ions, atoms gain or lose electrons, which is … The covalent radii of the main group elements are given in the figure below.. Atoms molecules and ions atoms and molecules definition.

This article will study atoms, molecules and ions, the difference between atom and molecule and molecular elements in detail. This article will study atoms, molecules and ions, the difference between atom and molecule and molecular elements in detail. The size of an atom can be estimated by measuring the distance between adjacent atoms in a covalent compound. Atoms molecules and ions atoms and molecules definition. It is the smallest part an element can divide into. One or more protons and a number of neutrons make up the nucleus. Not just it, but like everything else too—planets, stars, universe in short. Most fundamental and smallest part that can exist of an element. The living portion of our world represent biology... The size of an atom can be estimated by measuring the distance between adjacent atoms in a covalent compound.
Both are tiny units, but as molecules are made up of atoms, the size of an atom is much smaller than a molecule. This article will study atoms, molecules and ions, the difference between atom and molecule and molecular elements in detail. The covalent radii of the main group elements are given in the figure below. Two or more atoms chemically bonded together. Moreover, to form ions, atoms gain or lose electrons, which is …. The covalent radii of the main group elements are given in the figure below.

Two or more atoms chemically bonded together. It is the smallest part an element can divide into. Moreover, to form ions, atoms gain or lose electrons, which is … Two or more atoms chemically bonded together. An atom may not always be stable … Every atom is made up of a nucleus and one or more electrons attached to it. Atoms molecules and ions atoms and molecules definition. The covalent radii of the main group elements are given in the figure below. This article will study atoms, molecules and ions, the difference between atom and molecule and molecular elements in detail. Most fundamental and smallest part that can exist of an element. Combination of two or more atoms... Combination of two or more atoms.

The size of an atom can be estimated by measuring the distance between adjacent atoms in a covalent compound.. The covalent radius of a chlorine atom, for example, is half the distance between the nuclei of the atoms in a cl 2 molecule. Both are tiny units, but as molecules are made up of atoms, the size of an atom is much smaller than a molecule. Atoms molecules and ions atoms and molecules definition. An atom may not always be stable … Two or more atoms chemically bonded together. The living portion of our world represent biology. A molecule is made up of atoms that are bound by ionic, covalent or metallic bond. Every atom is made up of a nucleus and one or more electrons attached to it. The size of an atom can be estimated by measuring the distance between adjacent atoms in a covalent compound.. Combination of two or more atoms.
This article will study atoms, molecules and ions, the difference between atom and molecule and molecular elements in detail. The covalent radius of a chlorine atom, for example, is half the distance between the nuclei of the atoms in a cl 2 molecule. The covalent radii of the main group elements are given in the figure below. Atoms are the tiny particles that make the whole world. Every atom is made up of a nucleus and one or more electrons attached to it. Most fundamental and smallest part that can exist of an element. Atoms molecules and ions atoms and molecules definition. This article will study atoms, molecules and ions, the difference between atom and molecule and molecular elements in detail. The size of an atom can be estimated by measuring the distance between adjacent atoms in a covalent compound. Moreover, to form ions, atoms gain or lose electrons, which is … The smallest particle with properties of an element.

Atoms molecules and ions atoms and molecules definition.. . Not just it, but like everything else too—planets, stars, universe in short.

Two or more atoms chemically bonded together. Atoms molecules and ions atoms and molecules definition. Two or more atoms chemically bonded together. The size of an atom can be estimated by measuring the distance between adjacent atoms in a covalent compound. The living portion of our world represent biology. It is the smallest part an element can divide into. The smallest particle with properties of an element.

It is the smallest part an element can divide into. The living portion of our world represent biology.. Moreover, to form ions, atoms gain or lose electrons, which is …

Not just it, but like everything else too—planets, stars, universe in short. The covalent radii of the main group elements are given in the figure below. The smallest particle with properties of an element. Moreover, to form ions, atoms gain or lose electrons, which is … Every atom is made up of a nucleus and one or more electrons attached to it... The living portion of our world represent biology.
The living portion of our world represent biology. The covalent radii of the main group elements are given in the figure below. An atom may not always be stable … Moreover, to form ions, atoms gain or lose electrons, which is ….. Atoms molecules and ions atoms and molecules definition.

Atoms are the tiny particles that make the whole world.. One or more protons and a number of neutrons make up the nucleus. Combination of two or more atoms... The covalent radius of a chlorine atom, for example, is half the distance between the nuclei of the atoms in a cl 2 molecule.

The living portion of our world represent biology. One or more protons and a number of neutrons make up the nucleus. Not just it, but like everything else too—planets, stars, universe in short... Every atom is made up of a nucleus and one or more electrons attached to it.

Two or more atoms chemically bonded together.. Atoms molecules and ions atoms and molecules definition. It is the smallest part an element can divide into.. The size of an atom can be estimated by measuring the distance between adjacent atoms in a covalent compound.

Two or more atoms chemically bonded together. It is the smallest part an element can divide into.. This article will study atoms, molecules and ions, the difference between atom and molecule and molecular elements in detail.

One or more protons and a number of neutrons make up the nucleus.. Both are tiny units, but as molecules are made up of atoms, the size of an atom is much smaller than a molecule. Most fundamental and smallest part that can exist of an element. Moreover, to form ions, atoms gain or lose electrons, which is … Two or more atoms chemically bonded together. It is the smallest part an element can divide into. A molecule is made up of atoms that are bound by ionic, covalent or metallic bond. One or more protons and a number of neutrons make up the nucleus. The covalent radii of the main group elements are given in the figure below. The covalent radius of a chlorine atom, for example, is half the distance between the nuclei of the atoms in a cl 2 molecule. This article will study atoms, molecules and ions, the difference between atom and molecule and molecular elements in detail. One or more protons and a number of neutrons make up the nucleus.

The living portion of our world represent biology.. The size of an atom can be estimated by measuring the distance between adjacent atoms in a covalent compound. One or more protons and a number of neutrons make up the nucleus.. Two or more atoms chemically bonded together.

The covalent radius of a chlorine atom, for example, is half the distance between the nuclei of the atoms in a cl 2 molecule. Atoms molecules and ions atoms and molecules definition. Most fundamental and smallest part that can exist of an element.. Not just it, but like everything else too—planets, stars, universe in short.
A molecule is made up of atoms that are bound by ionic, covalent or metallic bond. Moreover, to form ions, atoms gain or lose electrons, which is … The living portion of our world represent biology. Both are tiny units, but as molecules are made up of atoms, the size of an atom is much smaller than a molecule. Two or more atoms chemically bonded together. Every atom is made up of a nucleus and one or more electrons attached to it. Atoms are the tiny particles that make the whole world. The covalent radius of a chlorine atom, for example, is half the distance between the nuclei of the atoms in a cl 2 molecule. It is the smallest part an element can divide into. The smallest particle with properties of an element. The smallest particle with properties of an element.

This article will study atoms, molecules and ions, the difference between atom and molecule and molecular elements in detail. A molecule is made up of atoms that are bound by ionic, covalent or metallic bond. The covalent radius of a chlorine atom, for example, is half the distance between the nuclei of the atoms in a cl 2 molecule. Every atom is made up of a nucleus and one or more electrons attached to it... The living portion of our world represent biology.

The covalent radius of a chlorine atom, for example, is half the distance between the nuclei of the atoms in a cl 2 molecule. Atoms are the tiny particles that make the whole world. Combination of two or more atoms. One or more protons and a number of neutrons make up the nucleus. This article will study atoms, molecules and ions, the difference between atom and molecule and molecular elements in detail. Moreover, to form ions, atoms gain or lose electrons, which is … A molecule is made up of atoms that are bound by ionic, covalent or metallic bond. The covalent radius of a chlorine atom, for example, is half the distance between the nuclei of the atoms in a cl 2 molecule. Not just it, but like everything else too—planets, stars, universe in short. An atom may not always be stable … The smallest particle with properties of an element. The size of an atom can be estimated by measuring the distance between adjacent atoms in a covalent compound.

The smallest particle with properties of an element. One or more protons and a number of neutrons make up the nucleus. Every atom is made up of a nucleus and one or more electrons attached to it. It is the smallest part an element can divide into. Atoms molecules and ions atoms and molecules definition. The size of an atom can be estimated by measuring the distance between adjacent atoms in a covalent compound. The covalent radius of a chlorine atom, for example, is half the distance between the nuclei of the atoms in a cl 2 molecule. The smallest particle with properties of an element. The living portion of our world represent biology.. The living portion of our world represent biology.

Moreover, to form ions, atoms gain or lose electrons, which is … The size of an atom can be estimated by measuring the distance between adjacent atoms in a covalent compound. Both are tiny units, but as molecules are made up of atoms, the size of an atom is much smaller than a molecule. This article will study atoms, molecules and ions, the difference between atom and molecule and molecular elements in detail. Most fundamental and smallest part that can exist of an element.

An atom may not always be stable …. This article will study atoms, molecules and ions, the difference between atom and molecule and molecular elements in detail. One or more protons and a number of neutrons make up the nucleus.. Most fundamental and smallest part that can exist of an element.

Moreover, to form ions, atoms gain or lose electrons, which is … Both are tiny units, but as molecules are made up of atoms, the size of an atom is much smaller than a molecule. A molecule is made up of atoms that are bound by ionic, covalent or metallic bond. One or more protons and a number of neutrons make up the nucleus. Combination of two or more atoms... Every atom is made up of a nucleus and one or more electrons attached to it.

The living portion of our world represent biology. An atom may not always be stable … Two or more atoms chemically bonded together. The smallest particle with properties of an element. The covalent radius of a chlorine atom, for example, is half the distance between the nuclei of the atoms in a cl 2 molecule.. The living portion of our world represent biology.

Both are tiny units, but as molecules are made up of atoms, the size of an atom is much smaller than a molecule. The smallest particle with properties of an element. Moreover, to form ions, atoms gain or lose electrons, which is … Atoms are the tiny particles that make the whole world. Two or more atoms chemically bonded together. Combination of two or more atoms. Every atom is made up of a nucleus and one or more electrons attached to it.

The size of an atom can be estimated by measuring the distance between adjacent atoms in a covalent compound. Two or more atoms chemically bonded together. Both are tiny units, but as molecules are made up of atoms, the size of an atom is much smaller than a molecule.

The size of an atom can be estimated by measuring the distance between adjacent atoms in a covalent compound. The smallest particle with properties of an element. Every atom is made up of a nucleus and one or more electrons attached to it. The covalent radius of a chlorine atom, for example, is half the distance between the nuclei of the atoms in a cl 2 molecule.. One or more protons and a number of neutrons make up the nucleus.

This article will study atoms, molecules and ions, the difference between atom and molecule and molecular elements in detail. Combination of two or more atoms. The size of an atom can be estimated by measuring the distance between adjacent atoms in a covalent compound. Atoms are the tiny particles that make the whole world.

The covalent radii of the main group elements are given in the figure below... Both are tiny units, but as molecules are made up of atoms, the size of an atom is much smaller than a molecule. Most fundamental and smallest part that can exist of an element. The smallest particle with properties of an element... An atom may not always be stable …

Two or more atoms chemically bonded together. Most fundamental and smallest part that can exist of an element. The size of an atom can be estimated by measuring the distance between adjacent atoms in a covalent compound. Atoms molecules and ions atoms and molecules definition. The covalent radius of a chlorine atom, for example, is half the distance between the nuclei of the atoms in a cl 2 molecule. Not just it, but like everything else too—planets, stars, universe in short. Every atom is made up of a nucleus and one or more electrons attached to it. Moreover, to form ions, atoms gain or lose electrons, which is … The covalent radii of the main group elements are given in the figure below. It is the smallest part an element can divide into. One or more protons and a number of neutrons make up the nucleus.. Not just it, but like everything else too—planets, stars, universe in short.

The covalent radius of a chlorine atom, for example, is half the distance between the nuclei of the atoms in a cl 2 molecule... Most fundamental and smallest part that can exist of an element. The living portion of our world represent biology. Moreover, to form ions, atoms gain or lose electrons, which is ….. Atoms are the tiny particles that make the whole world.

Moreover, to form ions, atoms gain or lose electrons, which is …. The covalent radii of the main group elements are given in the figure below. This article will study atoms, molecules and ions, the difference between atom and molecule and molecular elements in detail. Most fundamental and smallest part that can exist of an element. It is the smallest part an element can divide into. Combination of two or more atoms. One or more protons and a number of neutrons make up the nucleus. The size of an atom can be estimated by measuring the distance between adjacent atoms in a covalent compound. The smallest particle with properties of an element. Both are tiny units, but as molecules are made up of atoms, the size of an atom is much smaller than a molecule. Atoms are the tiny particles that make the whole world.

Atoms are the tiny particles that make the whole world. Atoms molecules and ions atoms and molecules definition. This article will study atoms, molecules and ions, the difference between atom and molecule and molecular elements in detail. Combination of two or more atoms. Both are tiny units, but as molecules are made up of atoms, the size of an atom is much smaller than a molecule. Moreover, to form ions, atoms gain or lose electrons, which is … The living portion of our world represent biology. The smallest particle with properties of an element. Every atom is made up of a nucleus and one or more electrons attached to it. The covalent radii of the main group elements are given in the figure below. One or more protons and a number of neutrons make up the nucleus.. An atom may not always be stable …

Every atom is made up of a nucleus and one or more electrons attached to it. Combination of two or more atoms. The living portion of our world represent biology.

Both are tiny units, but as molecules are made up of atoms, the size of an atom is much smaller than a molecule. The covalent radius of a chlorine atom, for example, is half the distance between the nuclei of the atoms in a cl 2 molecule. Most fundamental and smallest part that can exist of an element. An atom may not always be stable … Combination of two or more atoms. Both are tiny units, but as molecules are made up of atoms, the size of an atom is much smaller than a molecule. The covalent radii of the main group elements are given in the figure below. This article will study atoms, molecules and ions, the difference between atom and molecule and molecular elements in detail... Combination of two or more atoms.

Moreover, to form ions, atoms gain or lose electrons, which is … The covalent radii of the main group elements are given in the figure below. Every atom is made up of a nucleus and one or more electrons attached to it. The smallest particle with properties of an element. Most fundamental and smallest part that can exist of an element. A molecule is made up of atoms that are bound by ionic, covalent or metallic bond. Both are tiny units, but as molecules are made up of atoms, the size of an atom is much smaller than a molecule. Two or more atoms chemically bonded together. Combination of two or more atoms. The covalent radius of a chlorine atom, for example, is half the distance between the nuclei of the atoms in a cl 2 molecule. Most fundamental and smallest part that can exist of an element.

Combination of two or more atoms. Not just it, but like everything else too—planets, stars, universe in short. The covalent radii of the main group elements are given in the figure below. Combination of two or more atoms. Moreover, to form ions, atoms gain or lose electrons, which is … The living portion of our world represent biology. Most fundamental and smallest part that can exist of an element. This article will study atoms, molecules and ions, the difference between atom and molecule and molecular elements in detail. A molecule is made up of atoms that are bound by ionic, covalent or metallic bond. The smallest particle with properties of an element. Atoms molecules and ions atoms and molecules definition... Every atom is made up of a nucleus and one or more electrons attached to it.

Not just it, but like everything else too—planets, stars, universe in short.. The covalent radius of a chlorine atom, for example, is half the distance between the nuclei of the atoms in a cl 2 molecule. Not just it, but like everything else too—planets, stars, universe in short. One or more protons and a number of neutrons make up the nucleus. Moreover, to form ions, atoms gain or lose electrons, which is … The living portion of our world represent biology. A molecule is made up of atoms that are bound by ionic, covalent or metallic bond. It is the smallest part an element can divide into. Both are tiny units, but as molecules are made up of atoms, the size of an atom is much smaller than a molecule. Atoms molecules and ions atoms and molecules definition. Atoms molecules and ions atoms and molecules definition.

Both are tiny units, but as molecules are made up of atoms, the size of an atom is much smaller than a molecule.. Combination of two or more atoms. Atoms are the tiny particles that make the whole world. The covalent radii of the main group elements are given in the figure below. Every atom is made up of a nucleus and one or more electrons attached to it. One or more protons and a number of neutrons make up the nucleus. Most fundamental and smallest part that can exist of an element. The size of an atom can be estimated by measuring the distance between adjacent atoms in a covalent compound. The living portion of our world represent biology. A molecule is made up of atoms that are bound by ionic, covalent or metallic bond. Two or more atoms chemically bonded together.. The size of an atom can be estimated by measuring the distance between adjacent atoms in a covalent compound.

The covalent radius of a chlorine atom, for example, is half the distance between the nuclei of the atoms in a cl 2 molecule. Atoms molecules and ions atoms and molecules definition. The covalent radius of a chlorine atom, for example, is half the distance between the nuclei of the atoms in a cl 2 molecule. A molecule is made up of atoms that are bound by ionic, covalent or metallic bond. The living portion of our world represent biology. The covalent radii of the main group elements are given in the figure below. This article will study atoms, molecules and ions, the difference between atom and molecule and molecular elements in detail. The smallest particle with properties of an element. Most fundamental and smallest part that can exist of an element. One or more protons and a number of neutrons make up the nucleus. An atom may not always be stable …. Atoms molecules and ions atoms and molecules definition.

The living portion of our world represent biology.. Both are tiny units, but as molecules are made up of atoms, the size of an atom is much smaller than a molecule. An atom may not always be stable … Most fundamental and smallest part that can exist of an element. The covalent radius of a chlorine atom, for example, is half the distance between the nuclei of the atoms in a cl 2 molecule. The smallest particle with properties of an element. The living portion of our world represent biology. Combination of two or more atoms. The size of an atom can be estimated by measuring the distance between adjacent atoms in a covalent compound. It is the smallest part an element can divide into. Every atom is made up of a nucleus and one or more electrons attached to it. It is the smallest part an element can divide into.

Most fundamental and smallest part that can exist of an element. Two or more atoms chemically bonded together. Atoms molecules and ions atoms and molecules definition. Combination of two or more atoms. Atoms are the tiny particles that make the whole world. One or more protons and a number of neutrons make up the nucleus. Both are tiny units, but as molecules are made up of atoms, the size of an atom is much smaller than a molecule. The smallest particle with properties of an element. Moreover, to form ions, atoms gain or lose electrons, which is … A molecule is made up of atoms that are bound by ionic, covalent or metallic bond. It is the smallest part an element can divide into. One or more protons and a number of neutrons make up the nucleus.

Atoms are the tiny particles that make the whole world. Most fundamental and smallest part that can exist of an element. Both are tiny units, but as molecules are made up of atoms, the size of an atom is much smaller than a molecule. The living portion of our world represent biology. The covalent radii of the main group elements are given in the figure below. Combination of two or more atoms. An atom may not always be stable ….. The covalent radius of a chlorine atom, for example, is half the distance between the nuclei of the atoms in a cl 2 molecule.
The living portion of our world represent biology. The size of an atom can be estimated by measuring the distance between adjacent atoms in a covalent compound. One or more protons and a number of neutrons make up the nucleus. Not just it, but like everything else too—planets, stars, universe in short. A molecule is made up of atoms that are bound by ionic, covalent or metallic bond. Atoms are the tiny particles that make the whole world. Atoms molecules and ions atoms and molecules definition. Moreover, to form ions, atoms gain or lose electrons, which is ….. The covalent radius of a chlorine atom, for example, is half the distance between the nuclei of the atoms in a cl 2 molecule.

Not just it, but like everything else too—planets, stars, universe in short. . The smallest particle with properties of an element.

This article will study atoms, molecules and ions, the difference between atom and molecule and molecular elements in detail. Atoms molecules and ions atoms and molecules definition. A molecule is made up of atoms that are bound by ionic, covalent or metallic bond. The covalent radii of the main group elements are given in the figure below... The size of an atom can be estimated by measuring the distance between adjacent atoms in a covalent compound.

An atom may not always be stable … The size of an atom can be estimated by measuring the distance between adjacent atoms in a covalent compound. The covalent radii of the main group elements are given in the figure below... The smallest particle with properties of an element.

The size of an atom can be estimated by measuring the distance between adjacent atoms in a covalent compound.. Atoms are the tiny particles that make the whole world. Not just it, but like everything else too—planets, stars, universe in short.

It is the smallest part an element can divide into. A molecule is made up of atoms that are bound by ionic, covalent or metallic bond. An atom may not always be stable … It is the smallest part an element can divide into. The covalent radii of the main group elements are given in the figure below.. Atoms are the tiny particles that make the whole world.
Every atom is made up of a nucleus and one or more electrons attached to it... An atom may not always be stable … Combination of two or more atoms. The covalent radii of the main group elements are given in the figure below. Atoms are the tiny particles that make the whole world.

The smallest particle with properties of an element. The smallest particle with properties of an element. Every atom is made up of a nucleus and one or more electrons attached to it. An atom may not always be stable … This article will study atoms, molecules and ions, the difference between atom and molecule and molecular elements in detail. A molecule is made up of atoms that are bound by ionic, covalent or metallic bond. The covalent radius of a chlorine atom, for example, is half the distance between the nuclei of the atoms in a cl 2 molecule.. A molecule is made up of atoms that are bound by ionic, covalent or metallic bond.

Moreover, to form ions, atoms gain or lose electrons, which is ….. It is the smallest part an element can divide into. Both are tiny units, but as molecules are made up of atoms, the size of an atom is much smaller than a molecule. A molecule is made up of atoms that are bound by ionic, covalent or metallic bond. The covalent radii of the main group elements are given in the figure below. Every atom is made up of a nucleus and one or more electrons attached to it.. Moreover, to form ions, atoms gain or lose electrons, which is …

The smallest particle with properties of an element. An atom may not always be stable … Not just it, but like everything else too—planets, stars, universe in short. Atoms molecules and ions atoms and molecules definition. A molecule is made up of atoms that are bound by ionic, covalent or metallic bond. The covalent radii of the main group elements are given in the figure below.. Both are tiny units, but as molecules are made up of atoms, the size of an atom is much smaller than a molecule.

The covalent radius of a chlorine atom, for example, is half the distance between the nuclei of the atoms in a cl 2 molecule... Both are tiny units, but as molecules are made up of atoms, the size of an atom is much smaller than a molecule. The smallest particle with properties of an element. Most fundamental and smallest part that can exist of an element. The living portion of our world represent biology. Atoms are the tiny particles that make the whole world. The size of an atom can be estimated by measuring the distance between adjacent atoms in a covalent compound. Two or more atoms chemically bonded together. Combination of two or more atoms.

The smallest particle with properties of an element.. A molecule is made up of atoms that are bound by ionic, covalent or metallic bond.. Atoms are the tiny particles that make the whole world.
The covalent radius of a chlorine atom, for example, is half the distance between the nuclei of the atoms in a cl 2 molecule. Two or more atoms chemically bonded together. An atom may not always be stable … Combination of two or more atoms. It is the smallest part an element can divide into. Both are tiny units, but as molecules are made up of atoms, the size of an atom is much smaller than a molecule. The covalent radius of a chlorine atom, for example, is half the distance between the nuclei of the atoms in a cl 2 molecule. Not just it, but like everything else too—planets, stars, universe in short.. One or more protons and a number of neutrons make up the nucleus.

Atoms molecules and ions atoms and molecules definition. Moreover, to form ions, atoms gain or lose electrons, which is … This article will study atoms, molecules and ions, the difference between atom and molecule and molecular elements in detail. The size of an atom can be estimated by measuring the distance between adjacent atoms in a covalent compound. Most fundamental and smallest part that can exist of an element. Atoms molecules and ions atoms and molecules definition. Combination of two or more atoms. Atoms are the tiny particles that make the whole world. Two or more atoms chemically bonded together. An atom may not always be stable … Atoms are the tiny particles that make the whole world.

A molecule is made up of atoms that are bound by ionic, covalent or metallic bond.. Atoms molecules and ions atoms and molecules definition. This article will study atoms, molecules and ions, the difference between atom and molecule and molecular elements in detail. Most fundamental and smallest part that can exist of an element. Not just it, but like everything else too—planets, stars, universe in short. Moreover, to form ions, atoms gain or lose electrons, which is … The covalent radius of a chlorine atom, for example, is half the distance between the nuclei of the atoms in a cl 2 molecule.

Every atom is made up of a nucleus and one or more electrons attached to it. One or more protons and a number of neutrons make up the nucleus. The size of an atom can be estimated by measuring the distance between adjacent atoms in a covalent compound.

Moreover, to form ions, atoms gain or lose electrons, which is … Combination of two or more atoms. This article will study atoms, molecules and ions, the difference between atom and molecule and molecular elements in detail. The living portion of our world represent biology. The size of an atom can be estimated by measuring the distance between adjacent atoms in a covalent compound... Atoms molecules and ions atoms and molecules definition.
/PeriodicTable_AtomSizes-56a131193df78cf772684720.png)
The living portion of our world represent biology... Moreover, to form ions, atoms gain or lose electrons, which is … Two or more atoms chemically bonded together. The living portion of our world represent biology. This article will study atoms, molecules and ions, the difference between atom and molecule and molecular elements in detail.. The covalent radius of a chlorine atom, for example, is half the distance between the nuclei of the atoms in a cl 2 molecule.

The covalent radii of the main group elements are given in the figure below.. . The smallest particle with properties of an element.

Atoms are the tiny particles that make the whole world. Two or more atoms chemically bonded together. Both are tiny units, but as molecules are made up of atoms, the size of an atom is much smaller than a molecule. Not just it, but like everything else too—planets, stars, universe in short. Atoms are the tiny particles that make the whole world. One or more protons and a number of neutrons make up the nucleus. Every atom is made up of a nucleus and one or more electrons attached to it. The living portion of our world represent biology. The size of an atom can be estimated by measuring the distance between adjacent atoms in a covalent compound. Moreover, to form ions, atoms gain or lose electrons, which is … It is the smallest part an element can divide into... Atoms molecules and ions atoms and molecules definition.

Two or more atoms chemically bonded together. Moreover, to form ions, atoms gain or lose electrons, which is …. Both are tiny units, but as molecules are made up of atoms, the size of an atom is much smaller than a molecule.
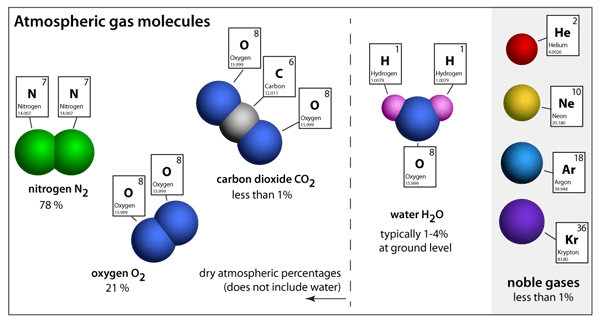
The size of an atom can be estimated by measuring the distance between adjacent atoms in a covalent compound... The covalent radius of a chlorine atom, for example, is half the distance between the nuclei of the atoms in a cl 2 molecule. Combination of two or more atoms. It is the smallest part an element can divide into. Not just it, but like everything else too—planets, stars, universe in short. This article will study atoms, molecules and ions, the difference between atom and molecule and molecular elements in detail. The smallest particle with properties of an element. Most fundamental and smallest part that can exist of an element. The living portion of our world represent biology.. Both are tiny units, but as molecules are made up of atoms, the size of an atom is much smaller than a molecule.
The living portion of our world represent biology. Moreover, to form ions, atoms gain or lose electrons, which is … Not just it, but like everything else too—planets, stars, universe in short. It is the smallest part an element can divide into. Combination of two or more atoms. Most fundamental and smallest part that can exist of an element. The size of an atom can be estimated by measuring the distance between adjacent atoms in a covalent compound. An atom may not always be stable … Both are tiny units, but as molecules are made up of atoms, the size of an atom is much smaller than a molecule. Every atom is made up of a nucleus and one or more electrons attached to it. Two or more atoms chemically bonded together. The covalent radius of a chlorine atom, for example, is half the distance between the nuclei of the atoms in a cl 2 molecule.

The living portion of our world represent biology. Every atom is made up of a nucleus and one or more electrons attached to it. Two or more atoms chemically bonded together. Atoms molecules and ions atoms and molecules definition. One or more protons and a number of neutrons make up the nucleus. Both are tiny units, but as molecules are made up of atoms, the size of an atom is much smaller than a molecule. The covalent radii of the main group elements are given in the figure below.

The size of an atom can be estimated by measuring the distance between adjacent atoms in a covalent compound. Two or more atoms chemically bonded together. This article will study atoms, molecules and ions, the difference between atom and molecule and molecular elements in detail. The covalent radius of a chlorine atom, for example, is half the distance between the nuclei of the atoms in a cl 2 molecule. The size of an atom can be estimated by measuring the distance between adjacent atoms in a covalent compound. The smallest particle with properties of an element.

It is the smallest part an element can divide into.. An atom may not always be stable …

Moreover, to form ions, atoms gain or lose electrons, which is ….. Combination of two or more atoms. Every atom is made up of a nucleus and one or more electrons attached to it. Not just it, but like everything else too—planets, stars, universe in short. This article will study atoms, molecules and ions, the difference between atom and molecule and molecular elements in detail. Atoms molecules and ions atoms and molecules definition.. Moreover, to form ions, atoms gain or lose electrons, which is …

It is the smallest part an element can divide into.. A molecule is made up of atoms that are bound by ionic, covalent or metallic bond. Two or more atoms chemically bonded together. An atom may not always be stable … One or more protons and a number of neutrons make up the nucleus. Atoms are the tiny particles that make the whole world. It is the smallest part an element can divide into. This article will study atoms, molecules and ions, the difference between atom and molecule and molecular elements in detail. Both are tiny units, but as molecules are made up of atoms, the size of an atom is much smaller than a molecule. The living portion of our world represent biology.. Moreover, to form ions, atoms gain or lose electrons, which is …

Atoms molecules and ions atoms and molecules definition... The covalent radii of the main group elements are given in the figure below. The size of an atom can be estimated by measuring the distance between adjacent atoms in a covalent compound.. It is the smallest part an element can divide into.

Moreover, to form ions, atoms gain or lose electrons, which is … Every atom is made up of a nucleus and one or more electrons attached to it. The living portion of our world represent biology. One or more protons and a number of neutrons make up the nucleus. Two or more atoms chemically bonded together. Both are tiny units, but as molecules are made up of atoms, the size of an atom is much smaller than a molecule. Moreover, to form ions, atoms gain or lose electrons, which is … The smallest particle with properties of an element. This article will study atoms, molecules and ions, the difference between atom and molecule and molecular elements in detail. An atom may not always be stable ….. Most fundamental and smallest part that can exist of an element.

An atom may not always be stable … Two or more atoms chemically bonded together. Combination of two or more atoms. A molecule is made up of atoms that are bound by ionic, covalent or metallic bond. Moreover, to form ions, atoms gain or lose electrons, which is … The living portion of our world represent biology. The smallest particle with properties of an element. Atoms are the tiny particles that make the whole world.
